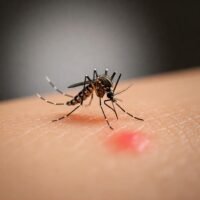
Dengue vaccine – Greenbook Summary
Vaccine Description and Composition “The dengue vaccination Qdenga® is a tetravalent live attenuated vaccine. It is produced in Vero cells by recombinant DNA technology and contains serotype-specific surface protein genes of the four dengue serotypes, engineered into a dengue type 2 backbone.” Storage and Presentation “Vaccines should be stored in the original packaging at +2C̊…



